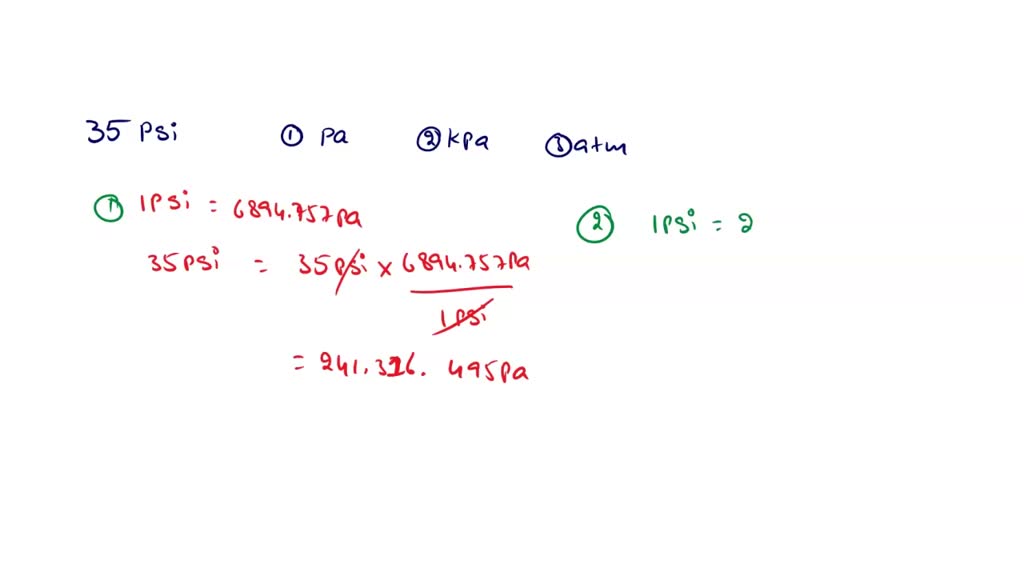
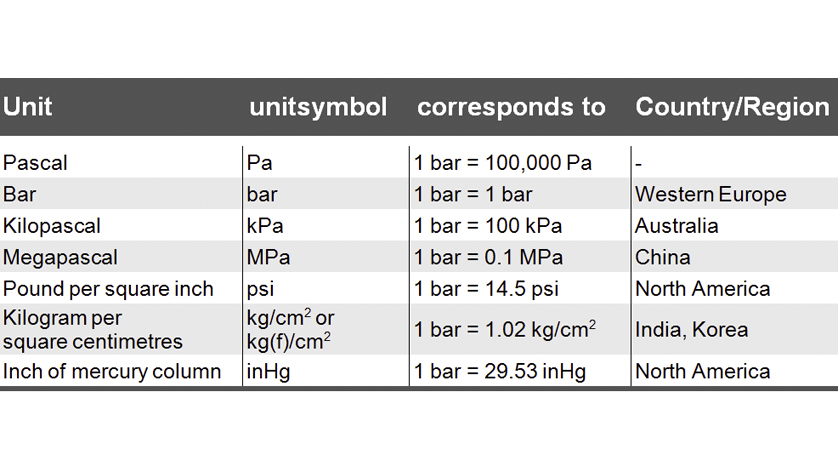
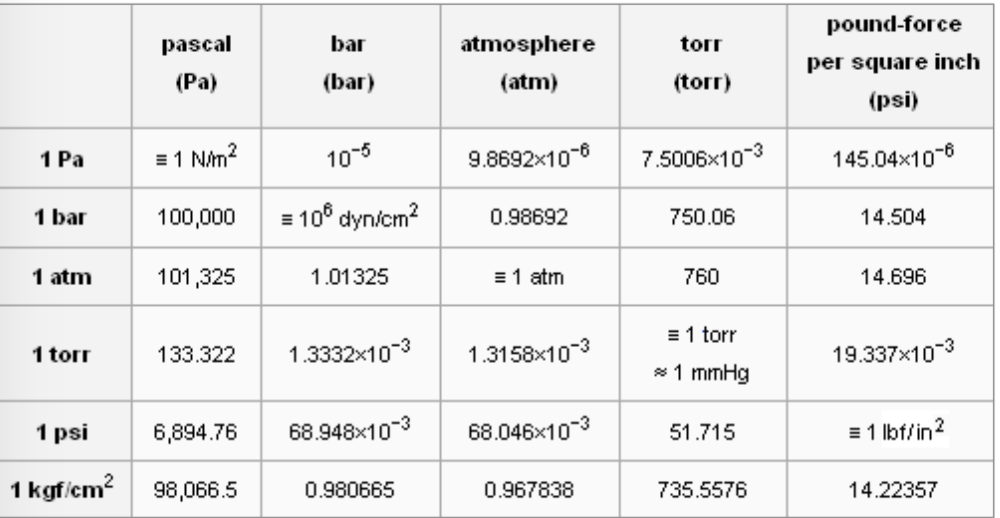
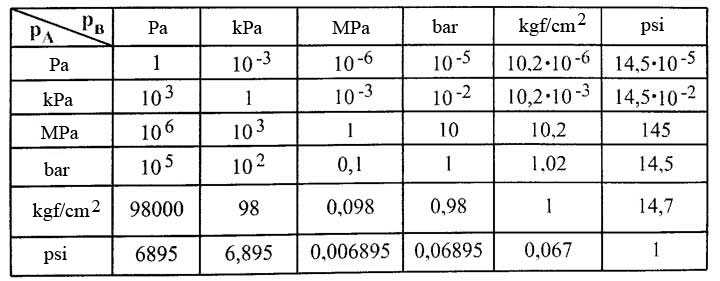
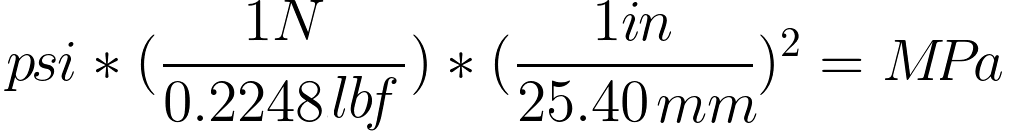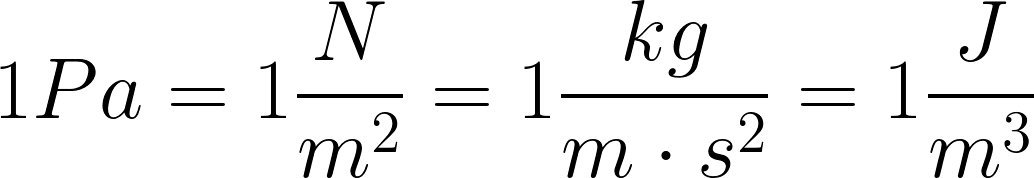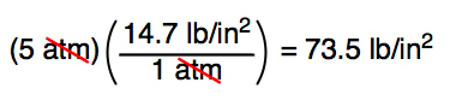SOLVED:Convert the following pressures to the SI unit of pascals (Pa), where 1 Pa=1 N / m 2^1 Pa=1 N / m^2: (a) 1500 kPa; (b) 35 psi; (c) 2.85 atm; (d) 883 torr.

Atmospheres and Conversions: 1.00 atm = x 10 5 Pa = kPa = 760. Torr = 14.7 psi To convert pressure: 1.Turn what you have to atm 2.Multiply. - ppt download